Purpose:
To provide evidence-based recommendations for the evaluation and diagnosis of endometriosis.
Target Population:
Reproductive-aged adults and adolescents with symptoms suggestive of endometriosis.
Methods:
This guideline was developed using an a priori protocol in conjunction with a writing team consisting of two specialists in obstetrics and gynecology and one specialist in reproductive endocrinology and infertility appointed by the American College of Obstetricians & Gynecologists’ (ACOG) Committee on Clinical Practice Guidelines–Gynecology. ACOG medical librarians completed a comprehensive literature search for primary literature within Cochrane Library, Cochrane Collaboration Registry of Controlled Trials, EMBASE, PubMed, and MEDLINE. The National Institute for Health and Care Excellence (NICE) evidence review on endometriosis diagnosis and management served as the evidence base for many of the clinical considerations. Studies that moved forward to the full-text screening stage were assessed by two authors from the writing team based on standardized inclusion and exclusion criteria. Included studies underwent quality assessment, and a modified GRADE (Grading of Recommendations Assessment, Development and Evaluation) evidence-to-decision framework was applied to interpret and translate the evidence into recommendation statements.
Recommendations:
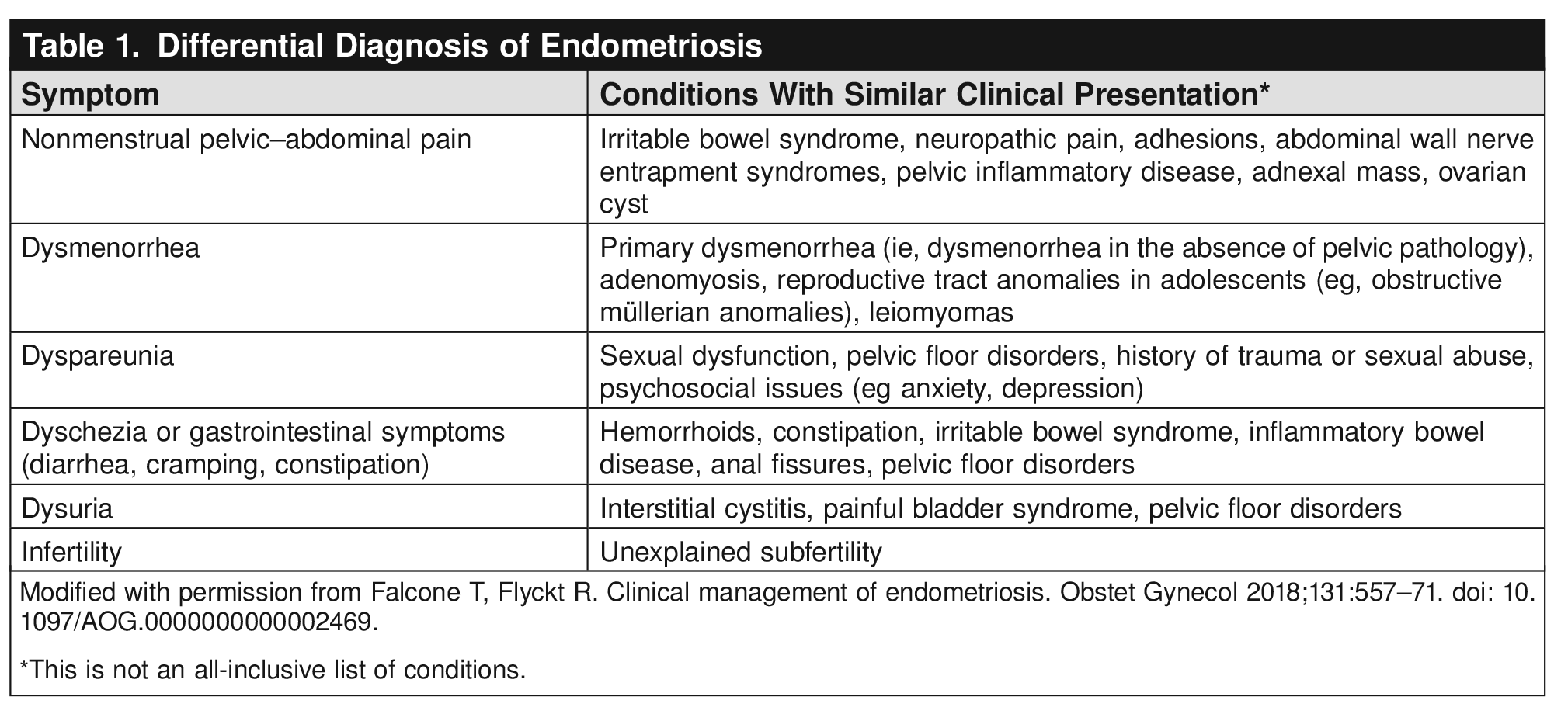
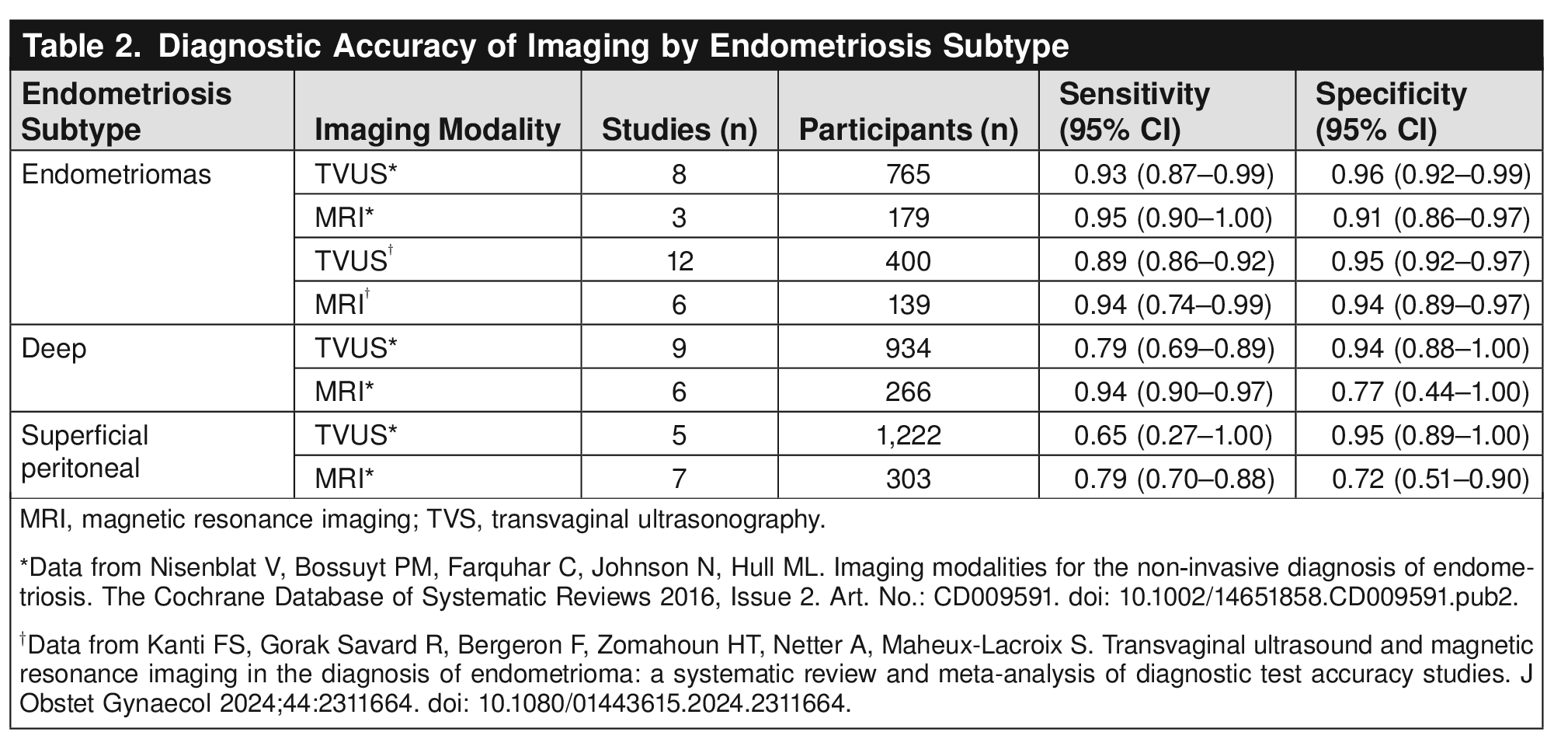
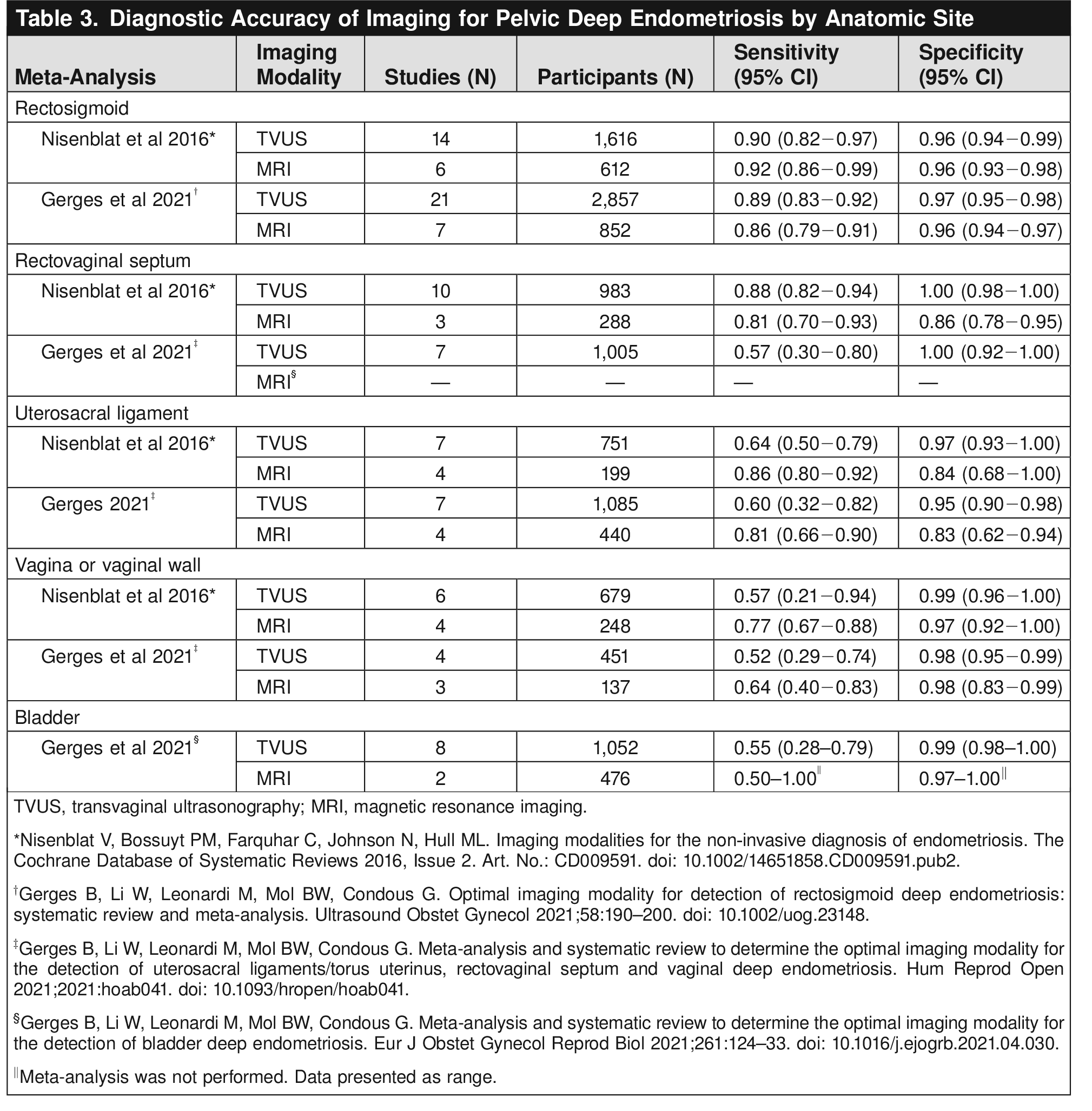
This Clinical Practice Guideline includes recommendations on the clinical, imaging, and surgical evaluation and diagnosis of endometriosis. Recommendations are classified by strength and evidence quality. Ungraded Good Practice Points are included to provide guidance when a formal recommendation cannot be made because of inadequate or nonexistent evidence. The recommendations included in this guideline also apply to adolescents unless otherwise specified and are based on review of the limited available evidence, extrapolated data from adult populations, and expert consensus.