Number 26 (Replaces Committee Opinion No. 741, June 2018; and Practice Advisory: Maternal Immunization, October 2022)
This Committee Statement was developed by the American College of Obstetricians & Gynecologists' Immunization, Infectious Disease, and Public Health Preparedness Expert Work Group in collaboration with Mark Turrentine, MD, Kevin A. Ault, MD, Rhoda Sperling, MD, and Laura Riley, MD.
ABSTRACT: Immunization is an essential part of preventive care for adults, including pregnant individuals. Each vaccine recommended for pregnant patients is important for the protection of the maternal–child dyad. Other vaccines provide maternal protection from severe morbidity related to specific pathogens such as pneumococcus, meningococcus, and hepatitis for at-risk pregnant individuals. Obstetrician–gynecologists and other obstetric care professionals should routinely assess their pregnant patients' vaccination status, including their risk factors for vaccine-preventable diseases. Based on this assessment they should recommend and, when possible, administer needed vaccines to their pregnant patients. There is no evidence of adverse fetal effects from vaccinating pregnant women with mRNA-derived vaccines, inactivated virus vaccines, bacterial vaccines, or toxoids. Real-world data continue to demonstrate the safety and efficacy of such use. Certain vaccines should be given in the postpartum period.
Summary of Recommendations and Conclusions
Based on the principles outlined in this Committee Statement, the American College of Obstetricians & Gynecologists (ACOG) makes the following recommendations:
Obstetrician–gynecologists and other obstetric care professionals should routinely assess their pregnant patients' vaccination status.
Obstetrician–gynecologists and other obstetric care professionals should recommend and, when possible, administer needed vaccines to their pregnant patients.
Individuals who are or will be pregnant during the fall/winter respiratory illness season should receive annual influenza and coronavirus disease 2019 (COVID-19) vaccines.
All pregnant individuals should receive a Tdap (tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis) vaccine during each pregnancy, as early in the 27–36 gestational-week window as possible.
All eligible pregnant individuals who meet criteria should receive the respiratory syncytial virus (RSV) vaccine.
Other vaccines may be recommended during pregnancy depending on the patient's age, prior immunizations, comorbidities, or disease risk factors.
Background
Immunization is an essential part of preventive care for adults, including pregnant individuals. Vaccination against infectious pathogens such as influenza, pertussis, coronavirus disease 2019 (COVID-19), and respiratory syncytial virus (RSV) is a proven safe and effective public health strategy 4. Each vaccine recommended during pregnancy is important for the protection of the maternal–child dyad. Other vaccines provide maternal protection from severe morbidity related to specific pathogens such as pneumococcus, meningococcus, and hepatitis for at-risk pregnant individuals. Since 2023, it has been recommended that pregnant patients aged 18 years or older be screened with a triple panel screen (HBsAg, anti-HBs, and total anti-HBc), if not previously completed, to identify persons who are susceptible and need vaccination 5. Adopting a comprehensive antepartum and postdelivery vaccine program will protect maternal health, reduce pregnancy complications, protect newborns and young infants, and help build community-wide protection. Obstetrician–gynecologists and other obstetric care professionals should routinely assess their pregnant patients' vaccination status, including their risk factors for vaccine-preventable diseases. An understanding of the primary indications for each vaccine recommendation, as well as the expected benefits and possible risks or contraindications, is needed to effectively counsel pregnant patients. Based on this assessment, obstetrician–gynecologists and other obstetric care professionals should recommend and, when possible, administer needed vaccines to their pregnant patients.
Influenza vaccination for pregnant individuals is critically important, because pregnant individuals with influenza are at greater risk of maternal morbidity and mortality in addition to fetal morbidity, including congenital anomalies, spontaneous abortion, preterm birth, and low birth weight 6. Influenza, Tdap (tetanus toxoid, reduced diphtheria toxoid, and acellular pertussis), COVID-19, and RSV vaccines provide fetal and neonatal benefit through passive transfer of protective antibodies across the placenta 4. There is no evidence of adverse fetal effects from vaccinating pregnant women with mRNA-derived vaccines, inactivated virus vaccines, bacterial vaccines, or toxoids, and a growing body of data demonstrate the safety of such use 4. Therefore, all pregnant individuals should receive annual influenza vaccination during influenza season, annual COVID-19 immunization, and Tdap vaccination with each pregnancy. Further, if eligible pregnant individuals meet criteria, administration of the RSV vaccine should be undertaken. Other recommended vaccines can be given in the postpartum period.
Disparities in Maternal Vaccination
Despite the recognized importance of vaccinations in preventing illness, uptake remains suboptimal and outbreaks of vaccine-preventable diseases currently are on the rise 7. Major challenges to increasing vaccine uptake and acceptance include widespread misinformation and disinformation on social media regarding safety; limited knowledge and awareness about recommended vaccinations; lack of trust in the medical system, especially in communities of color because of historic and ongoing injustices and systemic racism; prioritization of personal freedoms over collective health; and vaccination delay and refusal through nonmedical exemptions from state-mandated vaccination requirements.
The Role of Obstetrician–Gynecologists
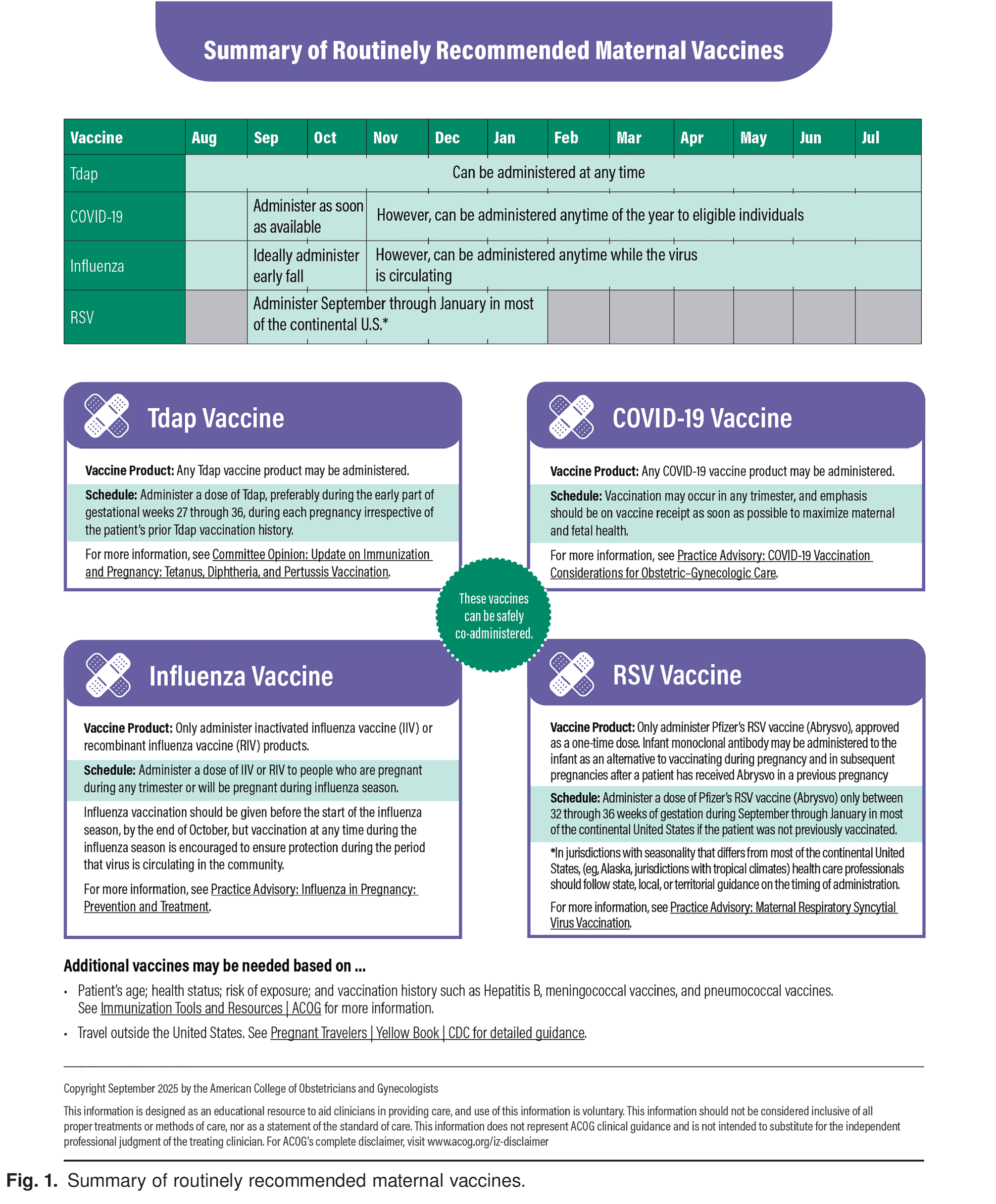
Obstetrician–gynecologists and other obstetric care professionals play a critical role in ensuring that pregnant individuals receive recommended vaccines and have the unique responsibility of counseling their patients through their vaccine decisions, including people who are pregnant and lactating 8. Despite the change in vaccine recommendations from the U.S. Department of Health and Human Services, the science has not changed Figure 1. It is recommended that obstetrician–gynecologists and other obstetric care professionals discuss these vaccines with their patients at their first prenatal encounter to develop a schedule that will optimize the time in gestation that immunizations will be given and to coordinate the need to administer multiple vaccines.

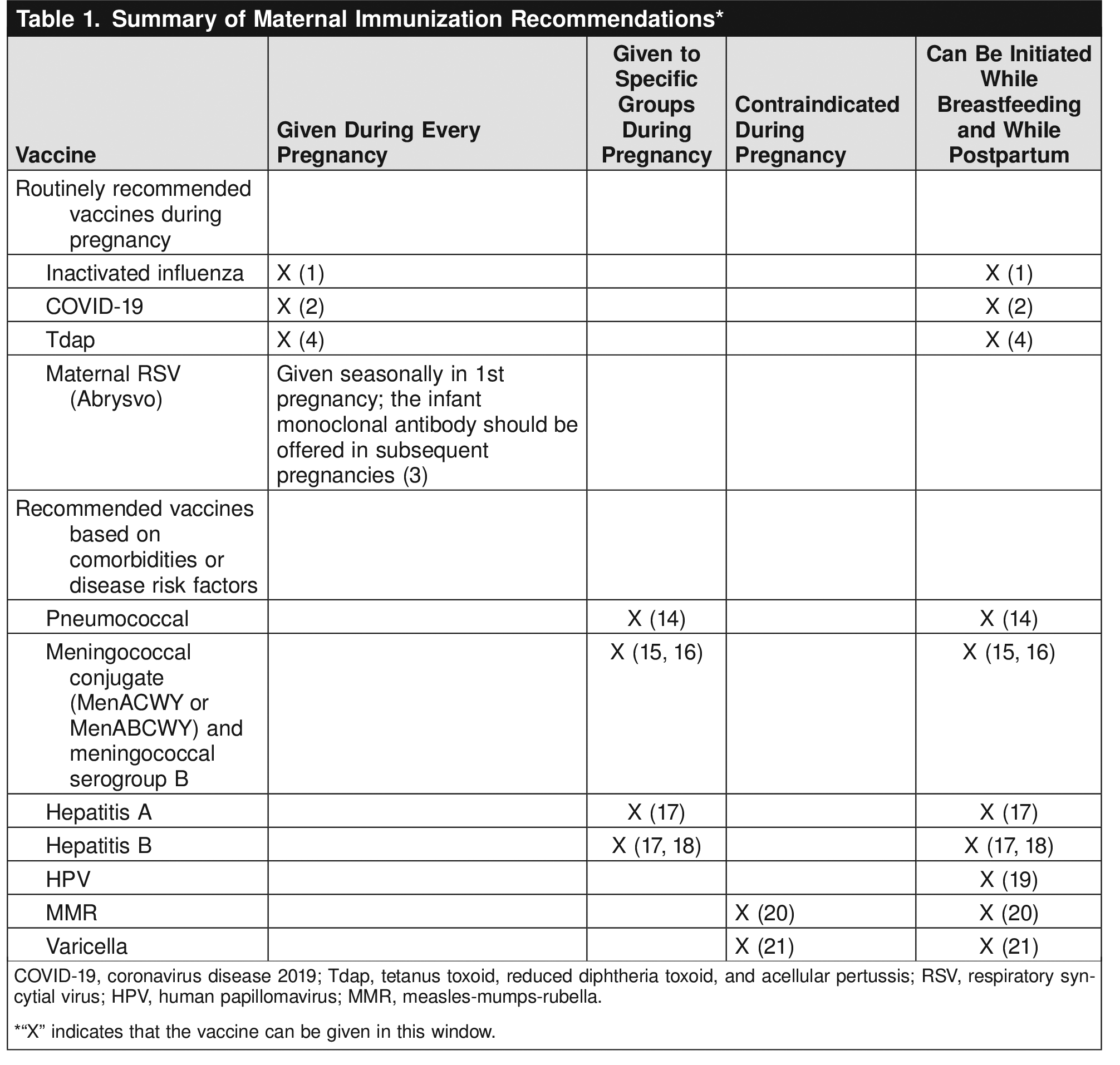
Studies consistently demonstrate that, when the recommendation and availability of vaccination during pregnancy comes directly from a woman's obstetrician or other obstetric care professional, the odds of vaccine acceptance and receipt are 5-fold to 50-fold higher 8 10 11 12 13. When communicating with patients, it is extremely important to provide a strong recommendation for vaccines as well as to underscore the general safety of vaccines and emphasize the fact that no steps were skipped in the development and evaluation of these maternal vaccines. As such, obstetrician–gynecologists and other obstetric care professionals should routinely assess their pregnant patients' vaccination status. Based on this assessment, they should recommend and, when possible, administer needed vaccines to their pregnant patients. Table 1 provides an easy-to-use reference to quickly assess which vaccines a pregnant individual needs and when they should be administered.

Individuals who are or will be pregnant during influenza season should receive annual influenza vaccine. Any of the licensed, recommended, age-appropriate inactivated influenza vaccines can be given safely during any trimester 4. A single dose of Pfizer's bivalent RSVpreF vaccine (Abrysvo), using seasonal administration, should be administered to eligible pregnant individuals to prevent RSV lower respiratory tract infection in infants Figure 1 Table 1. It is recommended that clinicians identify at-risk individuals who would benefit from other adult vaccines (for example pneumococcal vaccination). As part of antepartum care, susceptibility to vaccine-preventable diseases (eg, measles, rubella, varicella, and hepatitis B) should be assessed. As part of delivery planning, clinicians should recommend specific vaccines that should be administered postdelivery and advocate for the availability of these vaccines. Vaccines that may be required for travel are not included here. For information on travel vaccines during pregnancy, see https://www.acog.org/womens-health/faqs/travel-during-pregnancy.
Use of Language
The American College of Obstetricians & Gynecologists recognizes and supports the gender diversity of all patients who seek obstetric and gynecologic care. In original portions of this document, authors seek to use gender-inclusive language or gender-neutral language. When describing research findings, this document uses gender terminology reported by investigators. To review ACOG's policy on inclusive language, see https://www.acog.org/clinical-information/policy-and-position-statements/statements-of-policy/2022/inclusive-language.
Conflict of Interest Statement
All ACOG committee members and authors have submitted a conflict of interest disclosure statement related to this published product. Any potential conflicts have been considered and managed in accordance with ACOG's Conflict of Interest Disclosure Policy. The ACOG policies can be found on acog.org. For products jointly developed with other organizations, conflict of interest disclosures by representatives of the other organizations are addressed by those organizations.