This Practice Advisory was developed by the American College of Obstetricians & Gynecologists in collaboration with Samuel T. Bauer, MD, MBA, Nazanin Ahmadieh, DO, and Andrea D. Shields, MD, MS.
The American College of Obstetricians & Gynecologists (ACOG) endorsed* the Society for Maternal-Fetal Medicine Consult Series #74: Cell-free DNA screening for aneuploidies: Updated guidance 1 in November 2025. The guidance in Consult Series #74 and this Practice Advisory replaces ACOG Practice Bulletin No. 226, Screening for Fetal Chromosomal Abnormalities. 2
The Consult provides “updated guidance on the evolving applications of cell-free DNA (cfDNA) screening, including its use in detecting sex chromosome aneuploidies and microdeletions and its application in multifetal gestations.” 1 The guidance compares “cfDNA test performance with conventional screening methods and offers recommendations for managing inconclusive results and unexpected secondary findings.” 1 For information regarding prenatal diagnostic testing for genetic disorders, refer to Practice Bulletin No. 162, Prenatal Diagnostic Testing for Genetic Disorders. 3 For additional information regarding counseling about genetic testing and communicating test results, refer to Committee Opinion No. 693, Counseling About Genetic Testing and Communication of Genetic Test Results. 4 For information regarding carrier screening for genetic conditions, refer to Committee Opinion No. 690, Carrier Screening in the Age of Genomic Medicine, 5 and Committee Opinion No. 691, Carrier Screening for Genetic Conditions. 6 For information on ultrasound in pregnancy, including nuchal translucency measurement, refer to Practice Bulletin No. 175, Ultrasound in Pregnancy. 7
Summary of Recommendations
1,*,†
-
“We recommend that cfDNA screening for common aneuploidies (trisomies 21, 18, and 13) be made routinely available to all obstetrical patients (GRADE 1B).” 1
-
“We recommend cfDNA as the most sensitive and specific screening test for common fetal aneuploidies (trisomies 21, 18, and 13) in any patient population. After pretest counseling, every patient has the right to pursue or decline prenatal genetic screening and diagnostic testing (GRADE 1B).” 1
-
“We recommend that screening for sex chromosome aneuploidies be made available to obstetrical patients as an ‘opt-in’ consideration with appropriate pretest counseling (GRADE 1C).” 1
-
“We do not recommend routine general population screening for any microdeletion condition. Patients who choose to undergo cfDNA screening for 22q11.2 deletion specifically should do so only after appropriate pretest counseling. Pregnant people who are interested in obtaining information regarding the risk for fetal copy number variants should be offered diagnostic testing as opposed to cfDNA screening for microdeletion syndromes (GRADE 1C).” 1
-
“We recommend cfDNA as a first-line screening option for trisomy 21 detection in twin gestations (GRADE 1B).” 1
-
“Although the numbers of affected pregnancies are limited, the detection rates associated with trisomy 18 and 13 appear to be consistently high in twin gestations, and cfDNA screening for these conditions is recommended (GRADE 1B).” 1
-
“Because of a lack of data, cfDNA screening for sex chromosome aneuploidy in twin gestations and cfDNA screening for higher-order multiples are not recommended (GRADE 1C).” 1
-
“We do not recommend the routine use of cfDNA testing for large genome-wide copy number deletions or duplications (GRADE 1C).” 1
Components that should be included in pretest counseling for cfDNA screening, including an example, are covered in the endorsed guidance. 1
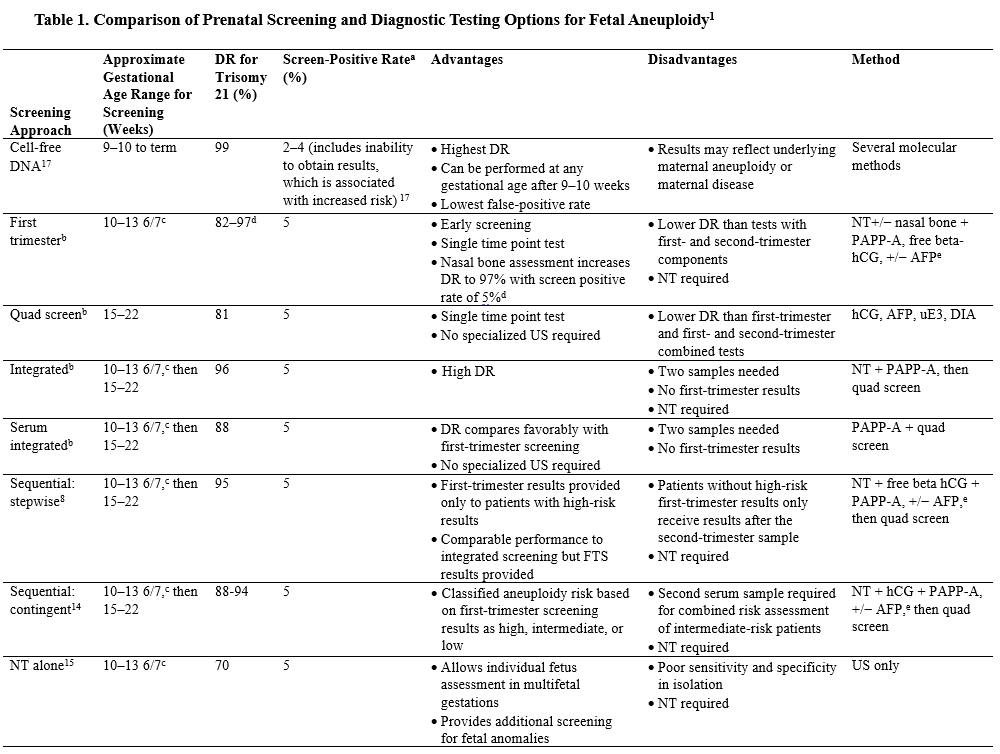
“Multiple studies have identified cfDNA as a superior screening test for trisomies 21, 18, and 13 compared with traditional serum and ultrasound screening tests Table 1 .” 1 8 9 10 11 Test performance for first- and second-trimester combined screening has a slightly lower detection rate for trisomy 21 than cfDNA screening. 12 13 14 15 Conventional combined serum or first-trimester ultrasound screening or both can lead to a range of findings that cfDNA may not detect, including fetal structural abnormalities, unanticipated genetic diagnoses, or risk for adverse perinatal outcomes. 1 2 When evaluating test options, it is crucial to consider factors such as test performance, availability, cost, personalized risk profiles, and patient information preferences.” 1
Although less frequently used since the uptake of cfDNA screening, first- and second-trimester serum analyte screening remains relevant if the patient prefers this screening after pretest counseling, or when cfDNA cannot be offered (eg, in cases of vanishing twin syndrome, maternal organ transplantation or stem cell therapy, maternal mosaicism or translocation, or lack of insurance coverage of cfDNA). In such situations, serum screening provides a noninvasive alternative and, in addition, may detect pathogenic deletions or duplications, single-gene disorders, and placental abnormalities not detected with cfDNA. “Laboratories remain available for first- and second-trimester serum analyses, and the Fetal Medicine Foundation offers nuchal translucency certification. 16 An anatomic survey is recommended for all patients to detect structural abnormalities regardless of aneuploidy screening method.” 1
Managing cfDNA Screening Test Results
†
“A positive cfDNA result should be followed by genetic counseling, a detailed anatomic survey, and a recommendation for diagnostic testing with chorionic villus sampling (CVS) or amniocentesis. […] Patients with nonreportable cfDNA results should be offered genetic counseling, comprehensive ultrasound evaluation, and diagnostic testing because of the increased risk for fetal aneuploidy. […] Whether the patient chooses to forgo diagnostic testing and instead reattempts cfDNA screening may depend on several factors, including the presence of abnormal ultrasound findings and gestational age.” 1

Reprinted with permission from Rink BD, Dugoff L, Kuller JA. Society for Maternal-Fetal Medicine Consult Series #74: Cell-free DNA screening for aneuploidies: updated guidance. Pregnancy 2025;1:e70139. doi: 10.1002/pmf2.70139. 1
Abbreviations: AFP, alpha-fetoprotein; DIA, dimeric inhibin-A; DR, detection rate; FTS, first-trimester screening; hCG, human chorionic gonadotropin; NPV, negative predictive value; NT, nuchal translucency; PAPP-A, pregnancy-associated plasma protein A; PPV, positive predictive value; uE3, unconjugated estriol; US, ultrasonography.
a A screen-positive test result includes true-positives and false-positives. For cell-free DNA, this includes the test failure rates given the association with increased risk of aneuploidy (see Gil et al 17 ).
b First-trimester combined screening: 87%, 85%, and 82% for measurements performed at 11 weeks, 12 weeks, and 13 weeks, respectively. 8
c Because of variations in growth and pregnancy dating, some fetuses at the lower and upper gestational age limits may fall outside the required crown–rump length range. Also, different laboratories use slightly different gestational age windows for their testing protocol.
d Use of free beta-hCG in conjunction with nasal bone assessment increases the DR to 97% with a screen-positive rate of 5%. 13
e Testing of first-trimester AFP depends on the commercial laboratory used. First-trimester AFP should not be used in lieu of second-trimester AFP for open fetal defects screening.
* Endorsement denotes that ACOG fully supports the clinical guidance in the document. Clinical documents endorsed by ACOG are considered official ACOG clinical guidance.
† Please read the ACOG-endorsed guidance in its entirety for full context, including an explanation of the Society for Maternal-Fetal Medicine’s grading of recommendations assessment, development, and evaluation (GRADE) system.
Please contact [email protected] with any questions.