Reaffirmed October 2025
This Practice Advisory was developed by the American College of Obstetricians and Gynecologists. The American College of Nurse-Midwives, the Association of Women’s Health, Obstetric and Neonatal Nurses, the National Association of Nurse Practitioners in Women’s Health, and the Society for Maternal-Fetal Medicine endorse this document.
Updated ACOG Recommendation
The American College of Obstetricians and Gynecologists (ACOG) continues to endorse the Centers for Disease Control and Prevention (CDC) Sexually Transmitted Infection Treatment Guidelines, 2021 1 . However, in the context of the rapidly increasing rates of congenital syphilis, obstetrician–gynecologists and other obstetric care professionals should screen all pregnant individuals serologically for syphilis at the first prenatal care visit, followed by universal rescreening during the third trimester and at birth, rather than use a risk-based approach to testing.
Rationale
Congenital syphilis cases are increasing at an alarming rate across the United States. From 2012 to 2021, congenital syphilis cases increased by 755%2 . In the United States, in 2022 alone, there were 3,755 cases of congenital syphilis. According to the CDC, 88% of congenital syphilis cases in 2022 could have been prevented with timely screening and treatment2.
Notably, two in five infants with congenital syphilis were born to people who did not receive any prenatal care2. Therefore, it is important to make any health care encounter during pregnancy—including those in emergency departments, jails, syringe service programs, and maternal and child health programs—an opportunity to screen for syphilis2.
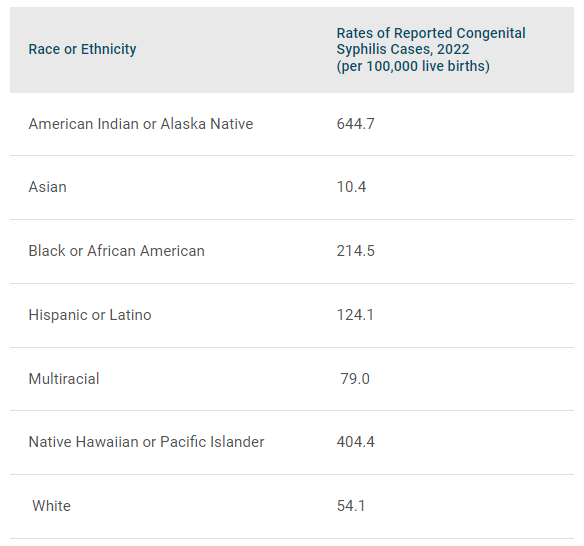
In addition, racial and ethnic inequities exist in congenital syphilis rates3 Table 1. It is important to acknowledge the harm and mistrust of the medical and public health establishment caused by the U.S. Public Health Service Untreated Syphilis Study at Tuskegee, which was conducted on a group of Black men—without collecting informed consent—and involved knowingly withholding syphilis treatment. It is necessary to approach our response to today’s congenital syphilis public health crisis with cultural awareness, humility, and sensitivity to build trust4.
Rates of Reported Congenital Syphilis Cases by Race/Hispanic Ethnicity of the Mother, United States, 2022

Data from Centers for Disease Control and Prevention. Sexually transmitted infections surveillance – Table 33, 2022. CDC; 2024. Accessed April 4, 2024. Available at https://www.cdc.gov/std/statistics/2022/tables/33.htm
Treatment of Syphilis in Pregnancy
Benzathine penicillin G is the only known effective treatment for syphilis in pregnancy and the prevention of congenital syphilis1. Timely initiation and completion of treatment are imperative and often complicated by stigma, multiple injections, treatment shortages, reporting and follow-up requirements, and mistrust of the medical system. In patients with a known severe allergy to penicillin, desensitization followed by penicillin treatment is recommended.
There is currently a shortage of benzathine penicillin G, sold as Bicillin L-A5. The U.S. Food and Drug Administration has exercised enforcement discretion for the temporary importation and use of Extencilline (benzathine benzylpenicillin injection, powder, for suspension) to mitigate the effects of the Bicillin L-A drug shortage. Extencilline is currently authorized and marketed in other countries. For more information on the use of Extencilline, read the Maternal Immunization Task Force Statement Obstetric Care Professionals Support the Availability and Use of Extencilline (Benzylpenicillin Benzathine) for the Treatment of Syphilis
When needed, local health departments, disease intervention specialists, and trusted community organizations can play an important role in helping people and their sex partners overcome barriers to accessing and completing treatment.
For additional information about syphilis in pregnancy and congenital syphilis, please see the following resources:
- Syphilis FAQs, Syphilis Screening in Pregnancy | ACOG
- Syphilis Screening Algorithm (PDF)
- Syphilis Treatment Among People Who are Pregnant in Six U.S. States – SET-NET, 2018-2021
- An Open Letter to Healthcare Providers on Syphilis Treatment and Prevention in 2023
- Labor of Love Podcast Season 3, Episode 2: Syphilis Surge: A Rising Concern in Pregnancy
- ACOG-Endorsed CDC Sexually Transmitted Infections Treatment Guidelines, 2021
- CDC Vital Signs: Syphilis in Babies Reflects Health System Failures
Please contact [email protected] with any questions.