Updated October 2025
This Practice Advisory was developed by the American College of Obstetricians and Gynecologists with the assistance of Naima T. Joseph, MD, MPH, Brenna L. Hughes, MD, and Laura E. Riley, MD.
From late 2023 – 2024, outbreaks of Oropouche were reported in several countries in South America and the Caribbean. On August 16, 2024, the Centers for Disease Control and Prevention (CDC) issued a Health Alert Network (HAN) Health Advisory to notify health care professionals, public health authorities, and the public about increased Oropouche virus activity, outside of areas where previously not found, and the potential risk during pregnancy of vertical transmission. Oropouche infections during pregnancy has been associated with microcephaly and other birth defects. Pregnant people are counseled to avoid non-essential travel to areas with active Oropouche transmission. Travel associated cases among U.S. residents have been reported, but local transmission has not been detected in the United States.
The American College of Obstetricians and Gynecologists is continuing to monitor the outbreak closely and will provide updated guidance as more information becomes available. The following is clinical guidance with unique considerations for pregnant individuals.
Oropouche virus, a member of the Orthobunyavirus genus, has been in known existence since 1955. The virus is mainly transmitted via bites from infected midges, although it can also be spread by mosquitoes. The virus has been primarily located in the Amazon basin. The 2024 Oropouche outbreak was associated with Oropouche virus disease in several countries in South America and the Carribean where Oropouche had not been previously identified. From 2024 – 2025, 109 cases from 7 U.S. states and territories of Oropouche diseases were voluntarily reported to the Centers for Disease Control. As of now, there is no evidence of local transmission in the United States 1 .
Clinical Presentation
The incubation period after exposure to Oropouche virus is typically 3–7 days. Symptoms are similar to dengue, Zika, and chikungunya, and usually involve abrupt fever, headache, chills, myalgias, and arthralgias. Other symptoms may include photophobia, dizziness, nausea and vomiting, and a maculopapular rash, starting on the trunk and spreading to the extremities. Severe symptoms, primarily neurological (including meningitis and meningoencephalitis), may also be present in a small percentage of patients. Symptoms usually last 2–7 days but may recur days or weeks later in up to 70% of patients 12. Infected individuals may also be asymptomatic 26. Oropouche is typically not fatal; however, two deaths among over 8,000 cases have been recently reported 3. Additionally, there is a risk of neuroinvasive disease; at least three cases of Guillain-Barre syndrome were identified in people who had been diagnosed with Oropouche virus disease in Cuba and 2 of the 109 cases reported to the CDC had complication of neuroinvasive disease 127. Currently, the clinical manifestations of Oropouche appear to be similar in pregnant and nonpregnant people; however, it is not known whether pregnant individuals manifest more severe disease.
Summary of Cases Involving Pregnancy
The Pan American Health Organization (PAHO) issued alerts regarding cases in Brazil and Cuba of possible vertical transmission of Oropouche virus and associated adverse pregnancy outcomes, such as miscarriage, stillbirth, and cases of newborns with microcephaly and other brain anomalies detected via retrospective investigation 428. In one case report involving a fetal demise, evidence of viral infection was present in fetal tissues, the umbilical cord, and the placenta 4. In those case reports involving microcephaly, retrospective analysis of serum and cerebrospinal fluid samples of newborns who had previously tested negative for dengue, chikungunya, Zika, and West Nile virus, were positive for OROV IgM. A limited case series of infants in Brazil born with microcephaly, arthrogryposis, and other congenital malformations without a confirmed cause identified six who tested positive for antibodies to Oropouche virus 26. It is important to note that data are limited and that these studies are insufficient to establish a causal relationship between Oropouche virus and adverse pregnancy effects. It is not known whether vertical transmission of Oropouche directly caused these fetal abnormalities, nor is the relationship between the timing of infection and pregnancy outcomes known.
Oropouche and Possible Sexual Transmission
There have been no reports of transmission of Oropouche virus through sexual activity to date. However, a recent publication describes a patient with Oropouche virus disease who had virus and viral RNA detected in bodily fluids, including semen. It is not known whether the virus can be transmitted through sex or if there is any clinical effect, but based on this recent development, the Centers for Disease Control and Prevention (CDC) published guidance for health care practitioners and patients on the possible sexual transmission of Oropouche (CDC sexual transmission HCP; CDC sexual transmission patient). This guidance includes a recommendation on condom use (summarized in the “Recommendations” section below).
Diagnosis
A suspected case of Oropouche involves an individual who has been in an area with documented or suspected Oropouche virus circulation within 2 weeks of initial symptom onset, and includes the following considerations:
-
Abrupt onset of reported fever, headache, and one or more of the following: myalgia, arthralgia, photophobia, retroorbital/eye pain, or signs/symptoms of neurological involvement (stiff neck, altered mental status, seizures, limb weakness, or cerebrospinal fluid pleocytosis), AND
-
Negative test results for other possible diseases, especially dengue, AND
-
Absence of a more likely clinical explanation 19.
Testing is recommended for both dengue and Oropouche because the clinical presentation is similar and clinical management of dengue can improve outcomes. Early supportive care is recommended for the treatment of dengue, because of its higher case fatality with severe disease 5.
Routine testing for Oropouche is not recommended if the patient does not have symptoms. However, even for patients without symptoms, healthcare providers can contact their local or state health departments for consultation if a pregnant person who traveled to an area with documented or suspected Oropouche, but has had no symptoms, and has fetal findings concerning for congenital Oropouche or experiences a stillbirth. Health departments will arrange testing after consultation with CDC. At this time, CDC is unable to perform testing on amniotic fluid or other pregnancy tissue 2919.
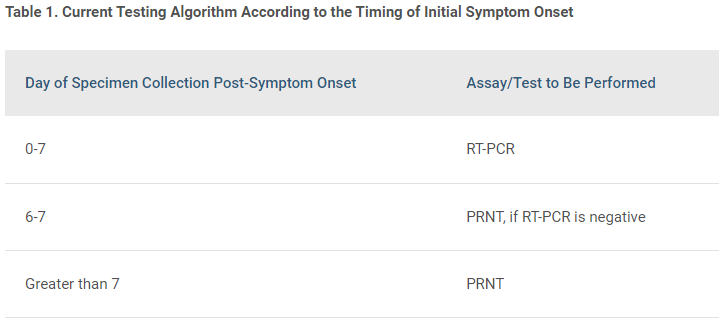
Current testing capabilities include CLIA-validated real-time reverse transcription–polymerase chain reaction (RT-PCR) to detect viral RNA and/or plaque reduction neutralization tests (PRNT) to detect virus-specific neutralizing antibodies in serum and/or cerebrospinal fluid 6. IgM antibodies usually appear toward the end of the first week of illness. To obtain testing for patients with an acute illness and risk factors (eg, recent travel), contact your local and state health departments to facilitate specimen collection through CDC’s Arbovirus Diagnostic Laboratory7. Specimens sent to the CDC will be tested according to the algorithm included below. Clinical diagnostic testing results will be sent to the public health partners, which can be shared with the treating clinician(s) 19.
The current testing algorithm, according to the timing of initial symptom onset, is as follows:

NOTE FOR PREGNANT PERSONS: If a specimen from a pregnant person is positive by PRNT only, acute and convalescent serum specimens, optimally collected 2 or more weeks apart, are preferred to confirm a recent infection by demonstrating a fourfold or greater change in neutralizing antibodies. Specimens collected within the first 2 weeks of infection are considered acute 19.
Prevention and Treatment
No antiviral treatments or vaccines are available for Oropouche. Supportive treatment includes rest, fluids, analgesics, and antipyretics. Hospitalization may be necessary for patients with more severe symptoms.
Pregnant individuals should be counseled regarding the possible risks to the fetus and newborn when considering travel to areas with reported Oropouche virus transmission. If travel is chosen, encourage use of personal protective measures, such as wearing long sleeves and pants and using Environmental Protection Agency (EPA)–approved insect repellant 8.
For pregnant patients with likely or confirmed diagnosis of Oropouche infection, fetal diagnostic testing is not currently available. Fetal evaluation via ultrasound, with particular attention to fetal neuroanatomy, should be performed in patients with confirmed disease when feasible. Pediatric providers should be notified of any clinical concerns to facilitate recommended infant testing and evaluation.
It is unknown whether Oropouche virus can be transmitted through human milk. However, given the benefits of breastfeeding or chestfeeding, it is still recommended in individuals with Oropouche infection.
Recommendations
Based on what is currently known, the following is recommended:
-
Counsel patients regarding the unknown risk for adverse pregnancy outcomes after Oropouche infection and to avoid travel to endemic areas. Refer to CDC resources regularly for updates on geographic exposure risk 921.
-
If travel to endemic areas is necessary, use appropriate precautions (using EPA-registered insect repellent, clothing to avoid bites, mosquito netting, etc.) 110.
-
For pregnant individuals who have been exposed in endemic areas and meet the criteria for suspected case definition within 14 days, contact the state or local health department to arrange for maternal testing (testing for both dengue and Oropouche)119.
-
For pregnant individuals without symptoms who traveled to an area with documented or suspected Oropouche and have fetal findings concerning for congenital Oropouche or experience a stillbirth, contact the state or local health department to arrange for maternal testing. At this time, CDC is unable to perform testing on amniotic fluid or other pregnancy tissue 29.
-
Treatment is supportive and includes rest, fluids, analgesics, and antipyretics.
-
There is no currently available diagnostic test to determine fetal infection. Fetal evaluation via ultrasound, with particular attention to fetal neuroanatomy, should be considered when feasible.
-
The CDC recommends considering serial fetal ultrasounds (every 4 weeks), adding that “in the absence of additional data, timing and frequency of ultrasounds can be individualized based on clinical judgement and patient circumstances; however, fetal findings may take time to manifest after infection" 11.
-
-
Suspected or confirmed Oropouche virus disease should be well documented in the prenatal care record to facilitate timely testing and treatment of the exposed infant 16.
-
Given the limited information on the risk of possible sexual transmission, any travelers to an area with a Level 1 or 2 Travel Health Notice for Oropouche and their partners who are concerned about possibly getting or passing Oropouche virus through sex, can consider using condoms during sex or not having sex during travel and for 6 weeks after returning from travel.
-
People assigned male at birth who are diagnosed with Oropouche, or who experienced symptoms compatible with Oropouche virus disease during or within 2 weeks of returning from travel to these areas, should be counseled to use condoms for at least six weeks following the symptom onset date.
-
The American College of Obstetricians and Gynecologists will continue to actively monitor the situation and update this Practice Advisory with additional information as needed. For more information, please refer to the following:
-
Health Advisory: Increased Oropouche Virus Activity and Associated Risk to Travelers | CDC 1
-
Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control | WHO 15
-
Interim Guidance for Evaluating and Managing Infants Born to Pregnant People with Confirmed or Probable Oropouche Virus Disease | CDC 16
-
Updated Interim Guidance for Health Departments on Testing and Reporting for Oropouche Virus Disease | Oropouche | CDC 19
-
Response to Oropouche Virus Disease Cases in U.S. States and Territories | Oropouche | CDC 20
-
Countries and Territories with Recent or Previous Oropouche Virus Transmission | Oropouche | CDC 21
-
Preventing Oropouche | Oropouche | CDC (for the general public) 23
-
Oropouche: Causes and How It Spreads | Oropouche | CDC (for the general public) 24
-
Oropouche Virus and Possible Sexual Transmission | Oropouche | CDC (for healthcare providers) 25
-
Interim Clinical Considerations for Pregnant People with Confirmed or Probably Oropouche Virus Disease 29
Please contact [email protected] with any questions.